Molecular Sciences Wageningen


Fervidobacterium gondwanense (F.g.), isolated from heated geothermal waters in Australia, is a member of the order Thermotogales. This bacterium grows between 44 °C and 88 °C, with an optimum at 68 °C. Its ability to grow on xylose indicates the presence of all genes involved in the xylose utilization pathway, including the gene encoding the xylose isomerase enzyme (XylA). This enzyme facilitates the conversion of D‑xylose into D‑xylulose as well as the conversion of D‑glucose into D‑fructose, which is desirable in many industrial processes.
The purpose of this research project was to isolate the previously undescribed xylA gene from Fervidobacterium gondwanense. We produced functionally active F.g. XylA protein in E. coli and biochemically characterized the purified protein.
Methods: A mini‑library containing F. gondwanense chromosomal DNA was created and screened using a Thermotoga maritima (T.m.) xylA ³²P‑ATP DNA probe. A positive restriction fragment of 3 kb was used to construct a recombinant vector, which was introduced into E. coli cells. The 3 kb insert from successfully transformed colonies was sequenced and was found to contain parts of the F.g. xylA gene. Further sequencing of deletion clones derived from the recombinant vector, together with the use of tailed‑primer PCR, revealed the complete F.g. xylA sequence. The acquired F.g. xylA was expressed in E. coli strain BL21 (DE3) in parallel with T.m. xylA, which served as control.
The F.g. XylA protein was isolated and purified, and D‑glucose isomerization activity was measured. Temperature optimum, pH dependency, and metal ion requirements were determined and compared with the enzyme properties of T.m. XylA. The F.g. xylA gene was sequenced and the genes surrounding the gene were identified. Comparison of the obtained sequence data with known xylA genes from related Thermotoga species showed up to 59% sequence identity. The deduced amino acid sequence of F.g. xylA was modeled using the known XylA structure of T. neapolitana, and both structures were compared with respect to the position of lysine residues that play an important role in protein deactivation at high temperatures and alkaline pH.
The XylA from F. gondwanense is a moderately thermophilic enzyme with optimal activity at 80 °C at slightly alkaline pH. The high amino‑acid homology with other Thermotoga XylA proteins makes this enzyme an excellent candidate for comparative studies on structure–function relationships.

L‑glutamate is the major excitatory neurotransmitter used in the vertebrate central nervous system. Glutamate released from presynaptic axon endings diffuses through the synaptic cleft and interacts with receptors located at dendritic spines of receiving neurons (model).
These interactions cause the opening of ion channels allowing Na+, K+ and Ca2+ ions to cross the postsynaptic membrane. The N‑methyl‑D‑aspartate (NMDA) receptor serves as an ion channel for Ca2+ ions, which act as secondary messengers activating proteins in the PostSynaptic Density (PSD).
Earlier experiments have shown that repeated stimulation induces changes in the levels of certain PSD proteins, strengthening postsynaptic responses. These phenomena are known as plasticity and Long Term Potentiation (LTP) and are believed to play a key role in memory and learning.
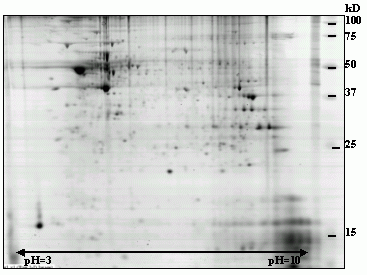
PSD proteins were analyzed using two‑dimensional electrophoresis (2‑DE). Synaptoneurosomes were isolated from rat brain tissue (SNS) and stimulated using KCl to induce glutamate release. Proteins were purified and separated using 2‑DE, and relative protein levels were quantified using PDQUEST software (Bio‑Rad).
A total of 238 protein spots were analyzed. We observed significant changes in 71 spots (p < 0.05, Student’s t‑test), with seven showing more than two‑fold regulation (example spot 8504). These results demonstrate regulation of a subset of proteins following stimulation, suggesting a functional link with LTP.